Introduction to Plain Tube Specifications for Blood Collection
When evaluating plain tube specifications for blood collection, technical assessors and procurement professionals must prioritize material safety, dimensional accuracy, and compatibility with centrifuges and microscopes. As a trusted medical device exporter, we understand the critical role of quality Centrifuge Tubes, Culture Tubes, and Microscope Slides in diagnostic accuracy. This guide helps distributors and lab specialists identify key parameters that ensure reliable sample integrity and workflow efficiency.
Key Parameters to Evaluate in Plain Blood Collection Tubes
Selecting the right plain tubes for blood collection requires a thorough understanding of critical specifications. Below are the essential factors to consider:
1. Material Composition
The tube material directly impacts sample integrity and chemical resistance. Borosilicate glass 3.3, for instance, offers superior thermal shock resistance and durability compared to standard soda-lime glass. This is particularly crucial when handling aggressive reagents or subjecting tubes to sterilization processes.
2. Dimensional Accuracy
Precise internal and external diameters ensure proper fit with centrifuge rotors and automated analyzers. Industry standards typically require:
- Wall thickness tolerance within ±0.2mm
- Length variation not exceeding ±1.5% of nominal size
- Roundness deviation below 0.5% of diameter
3. Graduation Markings
White enamel graduations should provide clear visibility with permanent markings that withstand repeated cleaning. The graduation intervals must comply with ISO 3819 standards for laboratory glassware.
Compatibility Considerations for Laboratory Workflows
Plain tubes must integrate seamlessly with existing laboratory equipment and processes. Key compatibility factors include:
For laboratories handling multiple sample types, consider Beaker alternatives when larger capacities (500–5000ml) are required for preliminary processing.
Quality Assurance and Certification Standards
Reputable manufacturers should provide comprehensive documentation verifying compliance with international standards:
Essential Certifications
- ISO 3819 for laboratory glassware
- USP Class VI for plastic components (if applicable)
- CE Marking for European market compliance
- FDA 21 CFR Part 820 for medical device manufacturing
Manufacturing Controls
Quality production processes should include:
- 100% visual inspection for defects
- Statistical process control for dimensional accuracy
- Batch traceability systems
- Validated sterilization methods (when applicable)
Procurement Decision Factors for Distributors
Medical distributors should evaluate suppliers based on both product specifications and supply chain reliability:
Frequently Asked Questions
How often should plain blood collection tubes be replaced in laboratory inventory?
Most laboratories implement a 12–18 month rotation cycle for glass tubes, while plastic variants may require more frequent replacement (6–12 months) due to potential polymer degradation. Always follow manufacturer recommendations and monitor for signs of wear.
What's the optimal tube size for routine blood collection?
For standard venipuncture, 3–5ml tubes (10–13mm diameter) offer the best balance between sample volume and handling convenience. Larger volumes (e.g., 50ml) may be needed for specialized tests or when using Beaker containers for initial sample processing.
How to verify tube compatibility with specific centrifuge models?
Always cross-reference the tube's maximum RCF rating with the centrifuge's specifications. Most manufacturers provide compatibility charts listing approved tube types for each rotor configuration.
Conclusion and Next Steps
Selecting optimal plain tubes for blood collection requires careful evaluation of material properties, dimensional accuracy, and workflow compatibility. By prioritizing these specifications, laboratories can ensure sample integrity while maintaining operational efficiency.
For procurement teams and distributors, establishing partnerships with certified manufacturers who provide comprehensive technical documentation and reliable supply chains is equally critical. Our expertise in medical device exports ensures access to quality-certified products that meet international standards.
Contact our technical specialists today to discuss your specific blood collection tube requirements and receive customized product recommendations for your laboratory or distribution network.
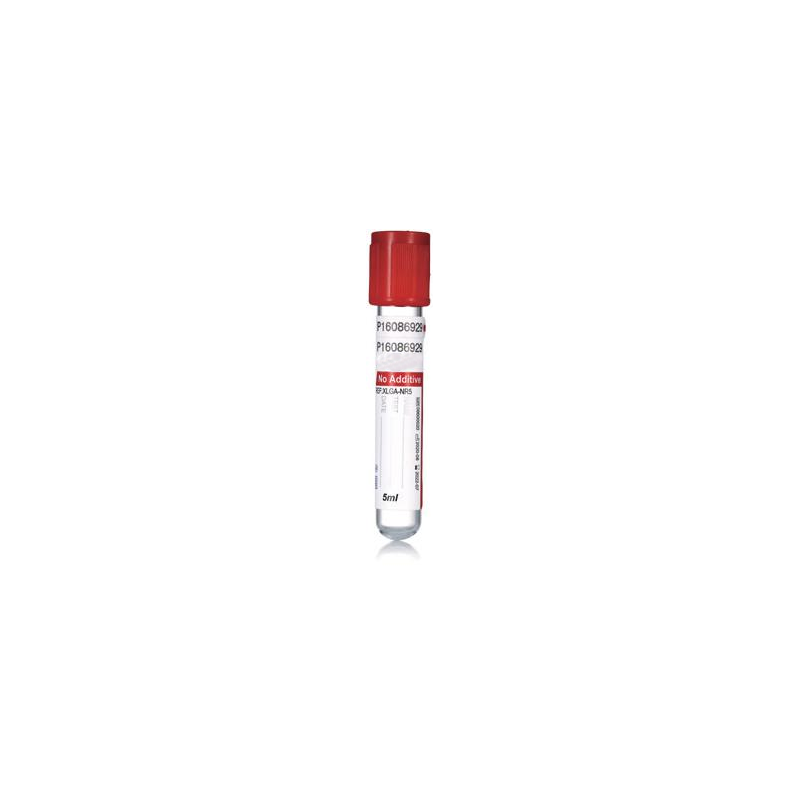
![Plain Tube Plain Tube]() Plain TubeVacuum Blood Collection Tubes are essential disposable devices for clinical blood collection. Designed with pre-set vacuum pressure, they draw accurate blood volumes safely and efficiently. Color-coded tops mark different additives for various tests. This closed system ensures sample quality, minimizes contamination, and meets standard laboratory requirements for blood testing and analysis.
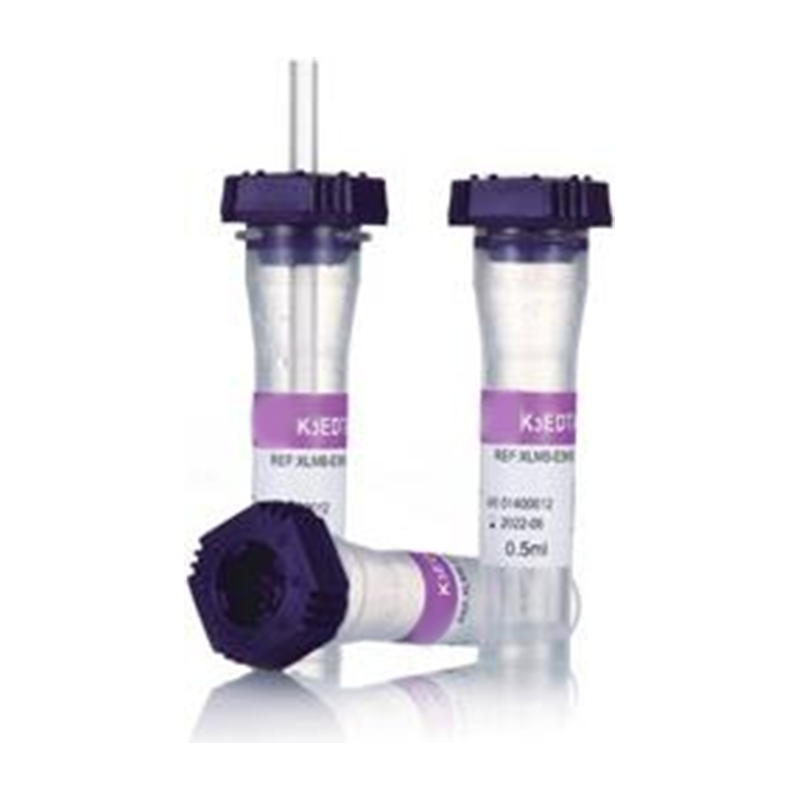
Plain TubeVacuum Blood Collection Tubes are essential disposable devices for clinical blood collection. Designed with pre-set vacuum pressure, they draw accurate blood volumes safely and efficiently. Color-coded tops mark different additives for various tests. This closed system ensures sample quality, minimizes contamination, and meets standard laboratory requirements for blood testing and analysis.![Mini Tube Type B Mini Tube Type B]() Mini Tube Type BMini Tubes are small-sized, disposable laboratory containers ideal for micro-sample storage and processing. Made of medical-grade plastic, they feature secure caps to prevent leakage and contamination. Compact and lightweight, they are widely used for storing small volumes of blood, reagents, and biological samples, ensuring safe preservation in clinical and laboratory settings.
Mini Tube Type BMini Tubes are small-sized, disposable laboratory containers ideal for micro-sample storage and processing. Made of medical-grade plastic, they feature secure caps to prevent leakage and contamination. Compact and lightweight, they are widely used for storing small volumes of blood, reagents, and biological samples, ensuring safe preservation in clinical and laboratory settings.![Positive Charged Slide Positive Charged Slide]() Positive Charged SlideThe Color-PLUSTM PCL series microscope slides additionally feature more hydrophobic surface and always come with a silky color coating at
Positive Charged SlideThe Color-PLUSTM PCL series microscope slides additionally feature more hydrophobic surface and always come with a silky color coating at
one surface End that is resistant to all common chemicals and routine stains that are utilized in laboratory.
Markable with the conventional labeling system, such as pencils, mark pens as well as most thermal transfer based slide printer, including
Thermo Slide-Mate® (Thermo Fisher) slide printer, , Signature® (Primera) slide printer and UV laser slide printer.
Recommended Application: manual IHC staining, automatic IHC staining with the Leica and Dako automated IHC Stainers and routine H&E
staining of detachable tissue sections
Related Posts
Online Message
PROFESSIONAL CONSULTATION
If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as we can.